
Common Names:
Lauri's Ring-barker
The Gregarious Phasmid
Identification:
 Head above, behind the antennæ, with several impressed longitudinal
lines, antennæ twenty-four jointed, about the length of the anterior
legs in the male, shorter in the female. Prothorax narrower than
the head, and becoming narrower to its junction with the mesothorax,
its length being about equal to the length of the head behind the
antennæ; the mesothorax is rather narrower and scarcely longer than
the prothorax except at its base, where it widens out; it is covered
beneath and on the sides with small tubercles, and on the back with a
double row of five larger tubercles; the metathorax is longer than the
mesothorax, and much wider ; it is sharply tuberculated beneath, as are
also, thouglh in a less degree, the abdominal segments. The tegmina are
rather pointcd - in the female half the length of the wings, in the male
about one-third. The wings are moderately large and equal in both sexes.
The legs are rather short; the hind femora strongly dentated beneath on
the inner and outer edge, with a deep groove between; the intermediate
femora are armed in the same way, but not so strongly; and the anterior
are grooved beneath, but not dentated. The basal joint of the tarsi is
a little the longest, except in the intermediate legs.
Head above, behind the antennæ, with several impressed longitudinal
lines, antennæ twenty-four jointed, about the length of the anterior
legs in the male, shorter in the female. Prothorax narrower than
the head, and becoming narrower to its junction with the mesothorax,
its length being about equal to the length of the head behind the
antennæ; the mesothorax is rather narrower and scarcely longer than
the prothorax except at its base, where it widens out; it is covered
beneath and on the sides with small tubercles, and on the back with a
double row of five larger tubercles; the metathorax is longer than the
mesothorax, and much wider ; it is sharply tuberculated beneath, as are
also, thouglh in a less degree, the abdominal segments. The tegmina are
rather pointcd - in the female half the length of the wings, in the male
about one-third. The wings are moderately large and equal in both sexes.
The legs are rather short; the hind femora strongly dentated beneath on
the inner and outer edge, with a deep groove between; the intermediate
femora are armed in the same way, but not so strongly; and the anterior
are grooved beneath, but not dentated. The basal joint of the tarsi is
a little the longest, except in the intermediate legs.
The specimens have been in spirits and therefore it is impossible to make
out the colouration with certainty, but the body seems to have been of a
reddish-brown, almost black beneath, the wing coverts yellowish, with the
median carina brown, the costal area of the wings brownish-yellow, and
the wings themselves hyaline, without any visible rosy tint. Length of
male three inches six lines; the female is not longer nor bulkier than
the male. This
uniformity of size in the sexes, if constant, is, I believe, quite
unprecedented in this family of insects.
(from Macleay,
1881)
the orange to brownish yellow base to the pre-anal part of the wing
(the coriaceous “costal field”); the shape of the operculum,
which is only very gently curved for most of its length, but sharply
so cephalad, and bears a few small laterocephalic teeth; and the
emarginate caudal margin of the poculum. (Key, 1957)
is distinguished
by the orange-yellow proximal patch on the remigium of the hind wing.
(from ?)
Length: 89mm
Coloring:
male and female volant
distinguished by the orange-yellow proximal patch on
the remigium of the hind wing.
Male:
Female:
In this image of a nymph, note the 2 rows of 4 bright red dots (short
spines) on the thorax. These are quite prominent on the nymph; they
are present but less prominent on the adult.
The black dot between the mid - and hind-wings is also prominent on
earlier instars, when the wings are less developed.
Lifespan: nypmhs take 3 months to mature,
the adult stage a further 3 months.
Eggs: dropped singly on to the forest floor beneath the crown of the
tree on which the female is feeding. Most of the eggs hatching in the
field are from fertilized females, but a small percentage may develop
parthenogenetically. The eggs lie amongst the forest litter for up
to eighteen months and sometimes longer before hatching. Generally no
development of the embryo is perceptible until the first summer following
oviposition, when development takes place though hatching is usually
delayed by a diapause until the next spring. In a small number of cases
the eggs develop and hatch within one year but some do not hatch until
the third season after oviposition.
Eggs from unmated females of P. wilkinsoni yielded female
individuals only, and thus the parthenogenesis appears to be thelytokous,
however it is rare (1.4%).
(from Campbell & Hadlington, 1967)
Nymphs hatch between late October and November, usually
during the early hours of the morning, if conditions at the time of
hatching are very dry or the egg is free to move, the nymph may be unable
to free its third pair of legs.
There are seven (male) or eight(female) nymphal instars,
growth usually being complete by mid-January.
(from Campbell & Hadlington, 1967)
Habitat:
arborial, top of host plants (not necessarity top of canopy)
Most of the Eucalyptus species are acceptable as food, though
there are preferences within the genus. While no quantitative tests
have been done on food preferences it has been noted in nature that
the narrow-leaved “peppermints” E. radiata Sieb. and
E. robersoni Blakely, the broad-leaved “peppermint”
E. dives Schauer and the “gums” E. viminalis
Labill., E. huberiana Naud., E. dairympleana Maiden,
E. mannifera (A. Cunn. Herb.) Mudie, E. stellulata
Sieb., E. pauciflora Sieb., and E. bicostata Maiden,
Blakely and Simmonds appear to be favoured species, and are the first
to be defoliated. Other species which are known to have
been severely defoliated are E. laevopinea R. T. Baker,
E. obliqua L'Hérit., E. delegatensis, R. T. Baker,
and E. fastigata Deane and Maiden, though these appear to be
less favoured than the former groups. E. andreana Naud. is an
acceptable species and has been used for most laboratory rearings.
Some Eucalyptus species are avoided and these will only be eaten
when there is no choice of food.
(Campbell & Hadlington)
Similar Species:
Rearing Notes:
Forrestry Commission of N.S.W reared this species for may years
while studying it.
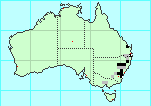 Range:
Range:
NE coastal, SE coastal, Murray-Darling basin, QLD, NSW
Status:
Not endangered. Known to occur in plage proportions from time to time.
Kentromorphic phase differences have been reported
(Key 1957) in the
Podacanthus wilkinsoni which sometimes reaches high population
densities. In the nymph, the pro-cryptic low-density phase is rather
uniform and usually green, whereas the conspicuous, supposedly aposematic,
high-density phase is patterned with black, yellow and sometimes white.
A mean density of one insect per eucalypt branchiet is sufficient to
induce the extreme high-density pattern, while the low- density extreme
occurs at less than one per 20 branchlets. Intermediate patterns appear
at intermediate densities, or in response to density change. The species
also shows morphometric phase differences analogous to those of locusts.
There is apparently no correlation between density and activity, and no
overt gregariousness.
(Key)
References
-
Balderson, J., Rentz,
D.C.F. and Roach, A.M.E. (1998).
in
Houston, W.K.K. & Wells, A. (1998) (eds)
Zoological Catalogue of Australia.
Vol. 23.
Archaeognatha, Zygentoma, Blattodea, Isoptera, Mantodea, Dermaptera,
Phasmatodea, Embioptera, Zoraptera.
Melbourne: CSIRO Publishing, Australia (ISBN 0643 06035 9).
pp. 347 - 376.
-
Bedford, G. O. (1970).
The development of the egg of Didymuria violescens
- embryology and determination of the stage at which first
diapause occurs.
Australian Journal of Zoology 18: 155-169
-
Bedford, G.O. (1978).
Biology and ecology of the Phasmatodea.
Ann. Rev. Entomol. 23: 125-149
-
Brewster, M.N., Brewster, A.A. and Crouch, N. (1946).
Family Phasmidæ (Leaf and Stick Insects).
In
Life Stories of Australian Insects.
Sydney: Lake & Ashes.
2nd Edition.
pp. 26-31
-
Campbell, K.G., 1961.
The effects of forest fires on three species of Stick Insects
(Phasmatidae Phasmatodea) occurring in plagues in forest areas of
south-eastern Australia.
Proceedings of the Linnaean Society of New South Wales,
86(1): 112-121
-
Campbell, K.G., 1966.
A review of research on disease and insects affecting forest trees in
Australia, New Zealand, and Oceania"
“Breeding pest-resistant trees”
Proceedings of a N.A.T.O. and N.S.F. Symposium
held at The Pennsylvania State University,
August 30 to September 11, 1964.
pp. 43-44.
Pergamon Press, 1966.
-
Campbell, K. G., Hadlington, P., 1967.
The biology of the three species of phasmatids which occur in
plague numbers in forests of south eastern Australia.
Forestry Commission NSW Res. Note No. 20, 38 pp.
-
Clark, J.T. (1976).
The eggs of stick insects (Phasmida): a review with
descriptions of the eggs of eleven species.
Syst. Ent. 1: 95-105.
-
Froggatt, W.W. (1905).
Notes on stick or leaf insects, with an account of Podacanthus
wilkinsoni as a forest pest, and the spiny leaf insect Extatosoma
tiaratum, in the orchard.
Agricultural Gazette of New South Wales 16: 515-520
-
Froggatt, W.W. (1923).
Forest Insects of Australia.
8vo. Sydney. Pp. i-viii,
1-171, 2 col. pls., 44 full pls., 33 text-blocks.
-
Goode, J. (1980).
Insects of Australia.
Angus & Robertson, Sydney.
ISBN 0 207 13427 8.
pp. 39-42
-
Hadlington, P.W., and Hoschke, F. (1959).
Observations on the ecology of the
phasmatid Ctenomorphodes tessulata (Gray).
Proceedings of the Linnaean Society of New South Wales.
84(2): 152-158
[I already have this one, except for plates 5 and 6, and possibly
page 159 (references). He also refers to figure 3 in the text, but none
appears in my copy; it may be on page 159.]
-
Hadlington, P.W. (1965)
Variations in diapause and parthenogenesis associated with
geographic populations of Podacanthus wilkinsoni Macl.
Journal of the Entomological Society of Australia, 2: 1-5
-
Hadlington, P.W., and Johnston, J.A. (1982).
An Introduction to Australian Insects.
University of New South Wales Press,
ISBN 0 86840 465 9,
pp. 28-29
-
Hughes, L., 1996.
When an Insect is more like a Plant.
Nature Australia, 25(4): 30-38
-
Key, K.H.L. (1970).
Phasmatodea (Stick-insects). pp. 394-404 in CSIRO (ed.) The
Insects of Australia. A textbook for students and research workers.
Melbourne: Melbourne University Press, Vol. 1, 1st Edn.
-
Kirby, W.F. (1904).
A Synonymic Catalogue of Orthoptera. 8vo. Vol. 1. Orthoptera,
Euplexoptera, Cursoria, et Gressoria (Forficulidæ, Hemimeridæ,
Blattidæ, Mantidæ, Phasmidæ). London: Longmans
& Co. x 501 pp.
-
Korboot, K. (1961)
Observations on life histories of
Acrophylla tessellata Gray and
Extatosoma tiaratum Macleay (Phasmida).
University of Queensland Papers Department of Entomology,
1(2): 161-69,
The University of Queeensland Pres, St. Lucia, 24th August.
-
Macleay, W.J. (1881).
On a species of Phasmatidæ destructive to Eucalipti.
Proceedings of the Linnaean Society of New South Wales,
3, December, 6: 536-539.
-
Musgrave, A. (1922).
Stick and Leaf Insects,
Australian Museum Magazine,
October, 1922, pp. 171-181
-
Rainbow, W.J. (1897).
Catalogue of the described Phasmidae of Australia.
Records of the Australian Museum, 3(2), 37-44.
[Note that he made a mistake re Extatosoma popa and E. tiaratum
according to
Gurney, A.B. (1947).
Notes on some remarkable Australasian walkingsticks, including a
synopsis of the Genus Extatosoma (Orthoptera: Phasmatidae).
Annals of the Entomological Society of America. 40(3): 373-396.
.]
-
Readshaw, J. L. (1965).
A theory of Phasmatid outbreak release.
Australian Journal of Zoology, 13: 475-90
-
Rentz, D.C.F (1996).
Grasshopper Country,
Chapter 16,
Phasmatodea: Leaf and Stick Insects,
pp. 244-257
-
Tepper, J.G.O. (1902).
List of the Described Genera and Species of the Australian and Polynesian
Phasmidæ (Spectre-Insects).
Transactions of the Royal Society of South Australia, 26: 278-287.
-
Tepper, J.G.O. (1903).
Notes and remarks on the described genera and species of Australian and
Polynesian Phasmidæ or ``spectre insects''.
Victorian Naturalist, 19: 138-142.
-
Vickery, V.R. (1983).
Catalogue of Australian stick insects (Phasmida,
Phasmatodea, Phasmatoptera, or Cheleutoptera). CSIRO
Australian Division of Entomology Technical Paper, No. 20, 15 pp.
- Search Google for
Podacanthus wilkinsoni,
or search Google Scholar for
Podacanthus wilkinsoni.
Copyright © 2000-2003
Peter Miller
This page was last changed 20-Sep-2006.
|

|

|


 Head above, behind the antennæ, with several impressed longitudinal
lines, antennæ twenty-four jointed, about the length of the anterior
legs in the male, shorter in the female. Prothorax narrower than
the head, and becoming narrower to its junction with the mesothorax,
its length being about equal to the length of the head behind the
antennæ; the mesothorax is rather narrower and scarcely longer than
the prothorax except at its base, where it widens out; it is covered
beneath and on the sides with small tubercles, and on the back with a
double row of five larger tubercles; the metathorax is longer than the
mesothorax, and much wider ; it is sharply tuberculated beneath, as are
also, thouglh in a less degree, the abdominal segments. The tegmina are
rather pointcd - in the female half the length of the wings, in the male
about one-third. The wings are moderately large and equal in both sexes.
The legs are rather short; the hind femora strongly dentated beneath on
the inner and outer edge, with a deep groove between; the intermediate
femora are armed in the same way, but not so strongly; and the anterior
are grooved beneath, but not dentated. The basal joint of the tarsi is
a little the longest, except in the intermediate legs.
Head above, behind the antennæ, with several impressed longitudinal
lines, antennæ twenty-four jointed, about the length of the anterior
legs in the male, shorter in the female. Prothorax narrower than
the head, and becoming narrower to its junction with the mesothorax,
its length being about equal to the length of the head behind the
antennæ; the mesothorax is rather narrower and scarcely longer than
the prothorax except at its base, where it widens out; it is covered
beneath and on the sides with small tubercles, and on the back with a
double row of five larger tubercles; the metathorax is longer than the
mesothorax, and much wider ; it is sharply tuberculated beneath, as are
also, thouglh in a less degree, the abdominal segments. The tegmina are
rather pointcd - in the female half the length of the wings, in the male
about one-third. The wings are moderately large and equal in both sexes.
The legs are rather short; the hind femora strongly dentated beneath on
the inner and outer edge, with a deep groove between; the intermediate
femora are armed in the same way, but not so strongly; and the anterior
are grooved beneath, but not dentated. The basal joint of the tarsi is
a little the longest, except in the intermediate legs.




 Range:
Range: 
